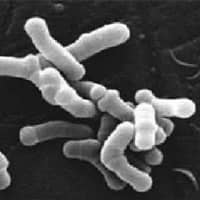
Bifidobacteria were discovered in 1899 in the faeces of breast-fed infants. Bifidobacteria is a genus of gram-positive, nonmotile (non-spore forming), anaerobic bacteria. They are ubiquitous in the gastrointestinal tract, vagina and mouth of mammals, including humans. Bifidobacteria are one of the major genera of bacteria that make up the gastrointestinal tract microbiota in mammals.
These bacteria are typically the most abundant species present in the intestine of breast-fed infants and are regarded as a primary reason for the infants’ greater resistance to disease. Bifidobacteria are recognised for their key role in the human intestinal microflora throughout life.
Healing Properties
- Probiotic: Bifidobacterium lactis (Bl-04) In vitro studies have shown that Bifidobacterium lactis (Bl-04) is extremely resistant to low pH conditions (which means it exhibits tolerance to the highly acidic conditions present in the stomach) and survives the presence of bile at concentrations present in the duodenum. This helps ensure that the probiotic microorganisms remain viable at the time of ingestion to allow them to confer their health benefits to the host. This implies that the probiotic survives GI tract passage and colonizes the host epithelium.[1]
Polyphenol-Responsive Bacteria
A 2026 systematic review and meta-analysis of human interventions reported that dietary polyphenol intake significantly increased Bifidobacterium abundance, with a pooled standardized mean difference (SMD) of 0.81 (95% CI: 0.18-1.44; p = 0.0114), indicating a moderate-to-large overall effect.[2]
Across 22 human studies, polyphenol-rich interventions were frequently associated with increases in Bifidobacterium, along with other beneficial genera such as Lactobacillus and Faecalibacterium.[2:1]
Bifidobacterium enrichment in these trials was linked to favorable shifts in gut ecosystem function, including improved intestinal barrier integrity, lower inflammation, and cardiometabolic improvements in some cohorts.[2:2]
The review notes that observed effects varied by polyphenol subclass, dose, intervention duration, baseline microbiome composition (metabotype), and the microbiome assessment method used.[2:3]
Mechanisms
Polyphenols are incompletely absorbed in the small intestine, so substantial amounts reach the colon where microbial metabolism converts them into smaller bioactive compounds that can shape microbial ecology, including Bifidobacterium-associated cross-feeding networks.[2:4]
Polyphenol-driven microbiome modulation is linked with increased short-chain fatty acid (SCFA)-related functionality, which may support host metabolic and immune homeostasis.[2:5]
Disease / Symptom Treatment
Dysbiosis-Related Conditions
In the 2026 human evidence synthesis, polyphenol-associated Bifidobacterium increases were observed alongside benefits reported in dysbiosis-associated conditions, including NAFLD, prediabetes, depression, and multiple sclerosis contexts.[2:6]
The authors emphasized that heterogeneity was substantial (I2 = 77.4%) and called for standardized long-term trials and multi-omics designs before stronger clinical conclusions are made.[2:7]
Study Type: Animal Study, Commentary, Human Study: In Vitro - In Vivo - In Silico, Human: Case Report, Meta Analysis, Review
Title:
Author(s):
Institution(s):
Publication:
Date:
Abstract:
Link: Source
Citations: ↩︎Title: Dietary Polyphenols as Modulators of Bifidobacterium in the Human Gut Microbiota
Publication: Nutrients
Date: 2026
Study Type: Systematic Review and Meta-Analysis (Human Intervention Studies)
Author(s): Corina Dalia Toderescu, Mohamudha Parveen, Svetlana Trifunschi, Alexandru Oancea, Gentiana Camelia Ciubuc Jurj, Iulia Gabriela Cresneac, Melania Florina Munteanu, Ioana Ciopanoiu, Casiana Boru, Elena Narcisa Pogurschi, Catalin Ionite, Alina Stefanache, Ionut Iulian Lungu
Copy: Source, PDF ↩︎ ↩︎ ↩︎ ↩︎ ↩︎ ↩︎ ↩︎ ↩︎